The journey from Real-World Data capture to Evidence-Based Decision Making involves a systematic process aimed at transforming raw data into actionable insights. This process is essential for creating a healthcare landscape that is responsive, effective and patient centric. But how can organizations achieve a full market understanding by harmonizing different types of data sources? And how does RWD help to enhance patient and HCP engagement?
In our Climedo Connect on March 13, 2024, Henrik Stork (Team lead Business Development, IQVIA) explored how to meaningfully leverage Real-World Data capture across the product lifecycle. We discussed:
- Ensuring maximum patient and HCP engagement with minimum effort and shared insights
- Combining retrospective data with longitudinal prospective data for maximum data quality and a 360° market understanding
-
Understanding various RWE use cases along the entire product lifecycle
In addition to Hendriks's presentation, attendees had the chance to take part in live polls, ask any questions they had and network with like-minded professionals.
Meet Our Speakers

Bradley Fryer (Webinar Host)
Business Development Manager
Climedo
As a Business Development Manager at Climedo, Brad accompanies pharma and medtech customers on their journey to successful clinical trials in both late and post-approval phases. He advises companies on the use of Climedo's modular, hybrid and integrated EDC & eCOA/ePRO solutions to save time, boost efficiency and bring the best treatments to patients faster. Brad has a PhD in Health Sciences and practiced for many years as a physiotherapist before moving into a commercially-focussed role.

Hendrik Stork
Associate Director BD
IQVIA
Hendrik is the Teamlead Business Development in IQVIA’s Real-World Solutions Team. With a strong background in value-based healthcare, RWE generation and especially Patient-Reported Outcomes, he has broad experience in cross-sectoral (inter)national healthcare projects and stakeholder management. He is also a lecturer at the University of Freiburg, supporting scientific innovation into application.
What was on the Agenda?
-
Intro (Bradley Fryer)
-
From Real-World Data Capture to Evidence-Based Decision Making (Hendrik Stork)
-
Discussion and networking (all)
- Wrap-up (Bradley Fryer)
Key Takeaways
INSIGHTS
Insights into strategies for achieving a comprehensive market understanding.
BEST PRACTICES
RWE use cases along the entire product lifecycle and understand the systematic process involved in transforming raw real-world data into actionable insights.NETWORKING & DISCUSSION
Connecting with other industry professionals and finding out how they are tackling similar challenges.
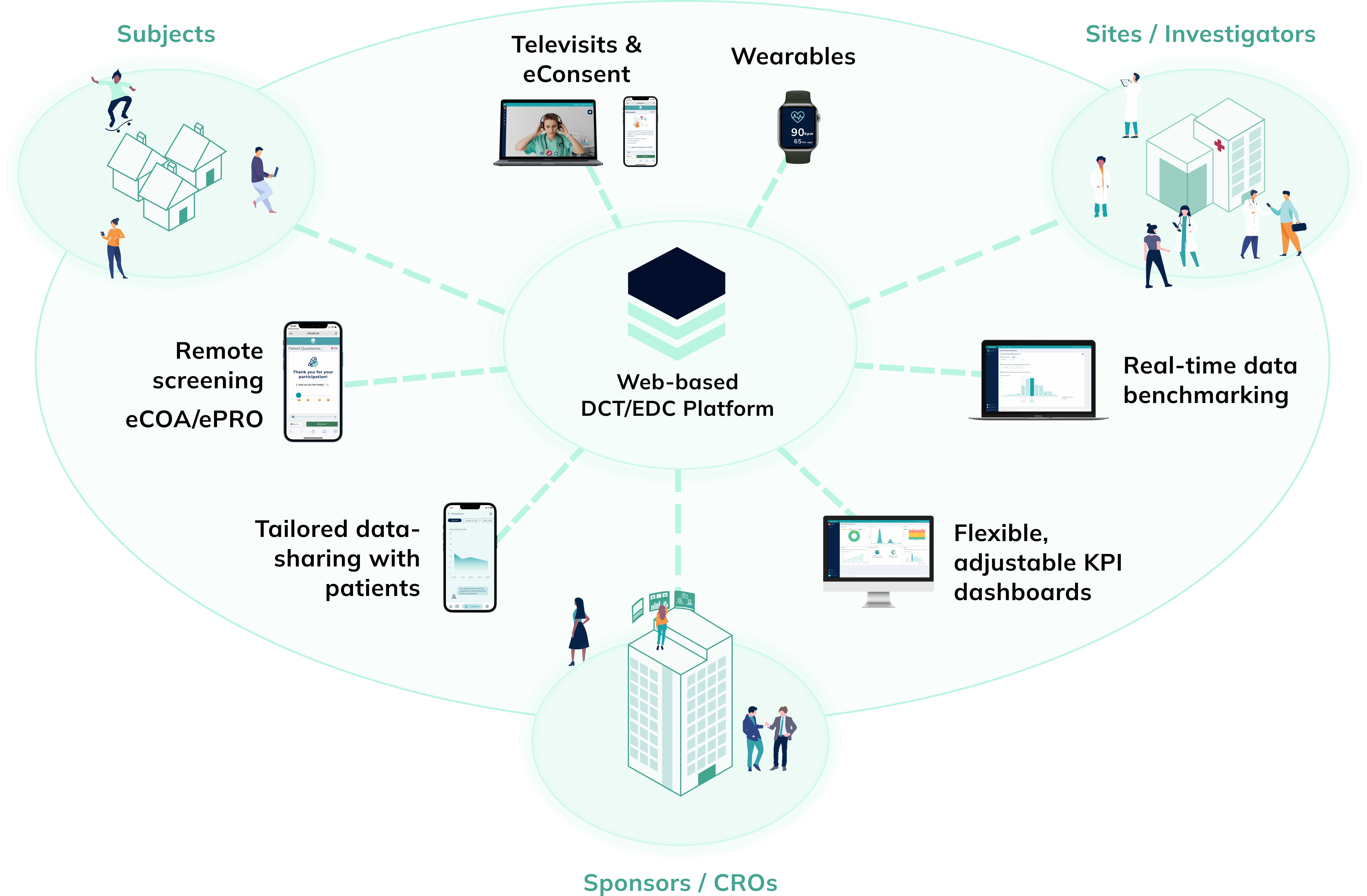
CLIMEDO
The leading European eCOA system for non-interventional studies, RWE and launch success
Climedo offers an all-in-one eCOA and EDC solution with hybrid capabilities for non-interventional studies and real-world evidence. By using a patient-centric approach and leveraging real-time data insights and visualizations around a study’s current progress, Climedo empowers its clients to better engage with healthcare professionals and other key opinion leaders (KOLs). This boosts awareness, stimulates scientific dialogue and accelerates the launch success of new medical innovations, thus reaching more patients faster.
Founded in Munich in 2017, Climedo is a leading trusted partner for pharma, medtech, CROs and academia with over 1.7 million patients enrolled to date. Learn more at www.climedo.com.